Manage your GxP processes in good quality with QMex's advanced features and modules.
What is QMex
It is a software designed modularly for the purpose of operating GxP processes electronically, which can be integrated in each module and other corporate applications, comply with 21 CFR Part 11 requirements and facilitates compliance with legal requirements that is accessed via internet browsers and validated according to GAMP 5.
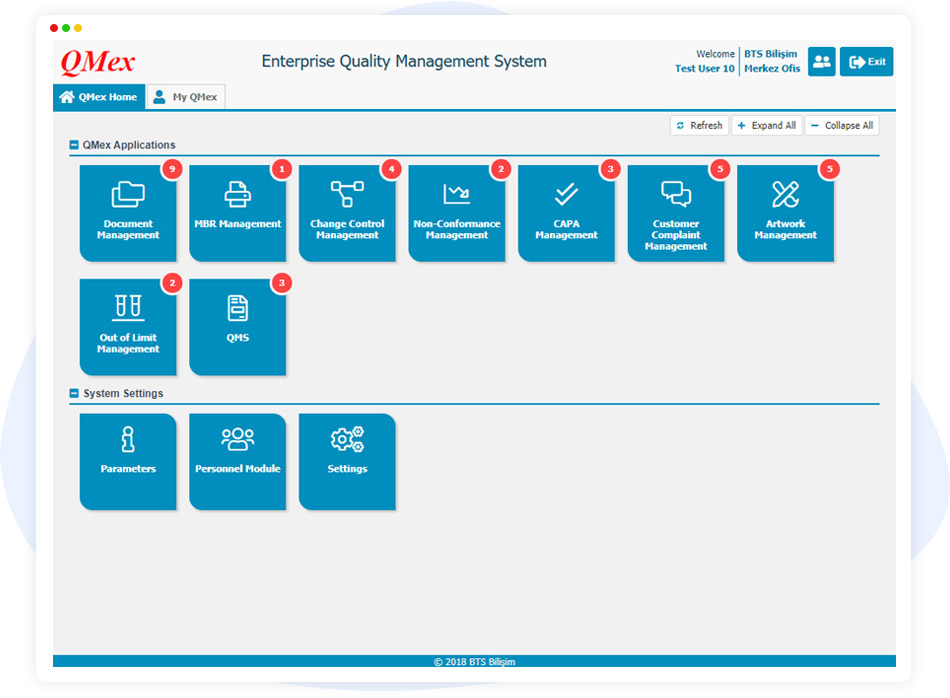
Powerful and safe key features of QMex
Secure
Data security is one of the key features of QMex. QMex has gained the trust of the market for years. Complies with 21 CFR Part 11 requirements.
Customizable
QMex is a ready-to-use product, it can be used immediately after the parameters are defined and validated after the installation. However, if desired, it can be customized according to your GxP processes.
Stable
As the data grows, it does not decrease performance, all functions work stably.
Validated
Validated according to GAMP 5. Validation documentation can be shared.
Flexible
Optionally, it can be licensed as cloud or on-premises.







